Preface. I was fortunate enough to know Walter for 15 years. He became a friend and mentor, helping me learn to become a better science writer, and sending me material I might be interested in, and delightful pictures of him sitting in a lawn chair and feeding wild deer who weren’t afraid of him. I thought his book Geodestinies: The Inevitable Control of Earth Resources over Nations and Individuals, published in 1997, was the best overview of energy and natural resources ever written, and encouraged him to write a second edition. He did try, but he spent so much time taking care of his ill wife, that he died before finishing it. I’ve made eight posts of just a few topics from the version that was in progress when he died at 96 years old in 2018 (500 pages).
More than any other energy and resources writer I know of, he focused on population as being the main problem that needed to be solved if we hoped to have a better future.
This first part covers many topics about energy, consumption, war, soil, oil production on various countries, and more. I have several more sections of this book in other posts under Experts/Walter Youngquist
Other Youngquist Geodestinies Posts:
- Walter Youngquist: Geodestinies
- Walter Youngquist: Geodestinies Coal
- Youngquist: the extraordinary geodestiny of Saudi Arabia and other gulf nations
- Youngquist on Oil, natural gas, heavy oil, tar sands, GTL, GTO, oil shale
- Walter Youngquist: Geodestinies Exponential growth
- Youngquist on Oil, natural gas, heavy oil, tar sands, GTL, GTO, oil shale
- Walter Youngquist: Geodestinies Exponential growth
- Walter Youngquist: Geodestinies Minerals
- Walter Youngquist: Geodestinies Metals
- Youngquist: Geodestinies Population
Alice Friedemann www.energyskeptic.com author of “Life After Fossil Fuels: A Reality Check on Alternative Energy“, 2021, Springer; “When Trucks Stop Running: Energy and the Future of Transportation”, 2015, Springer; Barriers to Making Algal Biofuels, and “Crunch! Whole Grain Artisan Chips and Crackers”. Podcasts: Collapse Chronicles, Derrick Jensen, Practical Prepping, KunstlerCast 253, KunstlerCast278, Peak Prosperity , XX2 report
***
The destinies of all nations and all people are in many ways bound up with the mineral and energy mineral resources of the Earth. Events of the geologic past have richly endowed some nations with valuable resources, whereas others have very few. The result is markedly different destinies for different nations. How these resources have affected the peoples of the past, how they influence our lives now, and how they will determine our futures is the study of GeoDestinies.
In a relative geological fraction of a second, these resources are being consumed. Soon these gifts from the past — in the case of mineral energy resources, petroleum, coal, and uranium — will be gone forever. Some metals can be reclaimed and recycled. Still, there is an inevitable percentage that will be dissipated and can never be recovered.
Currently in the United States, about 25 percent of all energy produced is used to produce other energy. Energy is essential to drill for oil, mine coal, mine uranium, cut wood, make solar and wind energy conversion devices, and so on. As the easily recoverable energy mineral resources (petroleum, coal, uranium) are increasingly exhausted, the cost in energy to produce more energy is estimated to rise to about 33% in the next ten years. It will almost certainly continue to rise after that. This trend in energy costs can be fatal. If it ultimately takes as much in energy as the energy produced by the effort, there is no energy surplus to use in other energy consuming sectors of the economy. At that point, the game is up.
Professor David Rutledge at the California Institute of Technology projects that 90% of all economically recoverable fossil fuels will be exhausted by 2070, meaning that they will be diminishing far earlier than that.
The globalization of commerce and trade by use of oil has led to the worldwide exploitation of mineral and energy resources by the industrialized and industrializing countries, in effect, “the tragedy of the commons” (Hardin, 1969) in its ultimate final form. The decline of oil production will in turn result in the return of local economies forced to exist on more locally available resources, as has been the circumstance during much of human history. This will greatly remake our economies and lifestyles from what we know today.
Due to differences in living standards, some populations (in industrialized countries) use more resources per capita than other populations. In some places local populations are so desperate to survive they destroy the environment with present use and cannot preserve it to sustain future population. In general, population growth and the environment are in direct conflict.
Around the world, the most easily discovered and recovered, higher-grade mineral and energy mineral deposits are used first. Thus, there is increased demand each year against resources that are declining in quality and cost more in both energy and money to obtain.
The desire for an understandable increased standard of living, together with the continuing rise in population, creates an exponential demand on mineral resources. In the first 50 years of the 20th century, the total world production of minerals and mineral fuels was far greater than the total production of these materials during all previous history. Then, in the following twenty years, this production was exceeded again by approximately 50%. When graphed, these statistics show an exponential curve that is rising steeply toward a vertical line. Such a rate of consumption cannot be sustained.
The dependence of the United States on imported minerals continues to increase. In 1986, the country imported 100% of five essential minerals. By 2000, the number of minerals for which we are 100% import-dependent increased to 13. U.S. dependence on imports includes manganese necessary for steel production, the yttrium used in color TVs and computer monitors, and mica used in electrical transformers. In 2000 the United States had a net import reliance of greater than 50% for 33 mineral materials.
Over the long-term, fertile soil and fresh water supplies of the United States are the most valuable of all its resources.
Minerals, including energy minerals, are the basis for our modern civilization. Nations not possessing these are either doomed to remain at a relatively low standard of living, or they have to get these resources in raw or finished form by trade or by moving industries abroad where the resources exist. Can free trade and access to raw materials prevent the “Japan decision” to go to war for resources? Minerals and energy minerals have markedly altered the course of civilization through warfare.
We must become stewards of the resources of our own regions, assuring that the fertility of the soil and the purity of water and air are maintained to provide material and energy needs and to absorb wastes, and recycling materials for continual use.
The story of the importance of minerals and the rise and progress of civilizations are parallel stories. The search for and discovery of minerals from salt to gold and silver have caused mass migrations of people. Searching for minerals has been the cause of the opening of new lands. It has been said that “the flag follows the miner’s pick.” The quest for gold and silver lured Spaniards to the New World, resulting in the conquests of Mexico, Colombia, Peru, and some of the adjacent lands.
Migrations of people today to resource areas, or to nations that can import resources, are perhaps greater than at any time in the past. Seeking jobs and a higher standard of living, largely based on Earth resources, people today are moving, both legally and illegally, by the millions.
As a result of resource depletion caused by rising population, refugees, often with different birthrates, will markedly change the demographics of much of the world in this century. The fundamental causes of resource-related population migration and growth, such as land degradation and desertification, energy, food and water demand, are the essential forces for monumental changes we will see this century (Duguid, 2004). People are on the move.
Although the resources are consumed mostly by the generations that discover and develop them, there is an argument that at least some of these riches should be passed on in some form to future generations who did not have the good fortune of living during this period of great mineral wealth. These are your children and your children’s children. To be sure, some of this wealth will be there in the future in the form of the buildings, factories, houses, roads, bridges, railroads, locks, canals, power stations, water lines, sewers, and many other structures for which mineral resources and the wealth derived from them are used. But in the case of fossil fuels, although they help to construct these physical features of civilization, their use leaves no legacy when used to power motor vehicles. The same is true with other energy uses such as space heating, and in producing products with a limited life such as automobiles, airplanes, household appliances, and sporting goods. Some of these products can be recycled, but inevitably much will be discarded.
The United States still has rich fertile land, but even that is being degraded by erosion and soil nutrient depletion, and the groundwater supplies needed to support irrigated lands are being over-pumped. Increasingly, the United States lives on “imported affluence.” Unending tides of immigrants continue to arrive in a land that is now the third most populous nation in the world, to share in a fading American dream.
In general, the oil we now recover is lower grade than previously. It takes more energy to refine the same high-quality end product from lower grade oil than higher-grade oil. Recovering oil from the Athabasca oil sands of Canada, for example, and then upgrading it to a product equal to what we can recover from conventional oil wells takes considerably more energy.
The answer to the question “what’s the answer?” is that it is not going to be possible to keep the current game going, hugely dependent as we are on fossil fuels. When I say this, I sometimes add that if I did have “the answer” and could patent it, within five years, I would be the world’s richest man. When I tell the audience there is no simple solution, I generally do not get invited back. We all like simple, inexpensive answers to problems. The problem of replacing oil offers no easy or inexpensive solutions.
The thin veneer of civilization. The isolated violence that occurred in the gasoline lines in the U.S. during the 1973 and 1979 oil crises suggest that civilization, even in a highly civilized United States, is a perilously thin veneer. The British social critic, C. P. Snow, wrote: “Civilization is hideously fragile and there’s not much between us and the horrors beneath, just about a coat of varnish.” That thin coat of varnish consists of the availability of the basics of life — food, shelter, and clothing. The ability to produce and distribute them in the huge quantities in which they are in demand today is possible only through the use of mineral and energy resources. Earth resources will increasingly affect the stability of our societies (LindahlKiesling, 1994).
Consumption
Since World War II, the world, led by the United States, has consumed more natural resources than in all previous history. That was the twentieth century.
North America, and particularly the United States, initially had resources in variety and abundance virtually unmatched by any other area of the world. With this marvelous spectrum of mineral and energy resources, the United States grew from a wilderness to become the richest and most powerful nation in the world in less than 400 years
Such development is historically unprecedented and can probably never be repeated.
By the late 1920s, the United States, with six percent of the world’s population, was producing 70 percent of the world’s oil, almost 50 percent of its copper, 46 percent of its iron, and 42 percent of its coal (Groner, 1972). The ability to produce and use these basic industrial materials and energy resources in large volumes was the key to the phenomenal development of the United States.
Including sand, gravel, cement, dimension stone, clay, and the energy and metal supplies already listed, more than five billion tons of new minerals are needed each year to support the U.S. economy. These demands add up to more than 20 tons of raw energy mineral and mineral supplies which have to be produced each year for every man, woman, and child in the United States, if our material standard of living is to be maintained. And, importantly, these demands increase in total size each year as our population grows by natural native increase, and by both legal and illegal immigration.
How fast a culture advances has largely been determined by what mineral and energy mineral resources it had available to it and to what degree technology development allowed them to be used. Mineral and energy resources combined with human ingenuity have been the mainsprings of civilization.
Energy is the key which unlocks all other natural resources and provides the physical and economic foundations of modern civilization (Ayers and Scarlott, 1952).
Without energy, the wheels of industry do not turn, no metals are mined and smelted. No cars, trucks, trains, ships or airplanes could be built, and if built, they could not move without energy. Without energy, houses would remain cold and unlighted; food would be uncooked. Our large agricultural areas could not be plowed or planted with the ease and on the vast scale they are today with relatively little human labor. Military defense as we know it today would not exist.
OIL
Pope (1996) commented, “If oil wealth is like winning the lottery, nations act like lottery winners: They tend to blow the money”. Some do, and some don’t.
Possibly the last mineral to significantly move people in the United States is the oil in the Bakken Formation in the Williston Basin of North Dakota. People from all across the United States moved to Williston to work in the development of the Bakken Field and all the supporting facilities. Building housing for the inflow of population could not meet demand, forcing people to sleep in their trucks. When the oil is gone, North Dakota will likely return to its agrarian economy, perhaps symbolic of all economies when the exploitation of nonrenewable Earth resources ends, and industrial societies must revert to agrarian economies.
Energy resources have since moved much of the population off the farms and into the cities. Today, with the use of petroleum powered farm machinery, only about two percent of America’s population farms the land, where a hundred years ago the majority of people did. But energy slaves (chiefly oil) now do the farm work, causing another great migration of people to the industrial areas, which, in turn, exist because of abundant energy to run factories.
Oil: A highly complex expensive enterprise. The oil industry plays an important part in countless ways in the lives of nearly all of us. The oil industry in its entirety including the exploration, drilling, production, transportation, refining, petrochemical production, and marketing uses more varied technology than any other industry in the world. These range from space satellites, to the highly complex science of organic chemistry. In between, seismology, directional drilling, drilling in waters a mile or more deep, and putting vast quantities of steam into the ground may be involved, among many other activities. The oil industry invests more money, by far, than any other industry in the world to conduct its operations.
WAR & RESOURCES
I wholeheartedly support Brown’s comments regarding military budgets and priorities. Brown observes that the projected military budget for the U.S. is $492 billion, which is approximately equal to the combined military budgets of all of the rest of the world combined. He proposes an annual U.S. budget of only $161 billion that would include such social goals as universal health care, family planning, adult literacy programs, and universal primary education. Goals and expenditures to benefit the Earth include reforestation, protecting topsoil on cropland, restoring fisheries, restoring rangelands, and protecting biological diversity.
The resource that all warring sides must have is water. Control of water supplies has been a tool in warfare in various ways. In the sixth century B.C., the King of Syria seized water wells as part of his campaign against Arabia. The Inca conquered the desert coastal cities of Peru such as Chan Chan by cutting off their water supply. In feudal times in Europe, castles or other fortresses under siege were vulnerable if they did not have water supplies within their walls. The enemy quickly determined if such was the case. Also, moats filled with water were part of the standard military protection of the day.
In peacetime, control of water can be used as a form of economic warfare. More than 30 nations receive one-third or more of their water from outside their borders. In the case of Egypt, it is 97 percent, Hungary 95 percent, Syria 79 percent, and Iraq 66 percent. With their surface waters chiefly under the control of other countries, these and 16 other nations which obtain more than 50 percent of their surface waters coming from outside their borders are vulnerable to economic water warfare.
Hitler’s oil supplies began to fail. German motorized divisions toward the end of the war suffered markedly from lack of oil. When General George Patton was finally on the move across France with the Germans in full retreat, pipeline specialists from Texas (where else!) followed Patton’s tanks and laid pipeline at a rate of up to 50 miles a day. And there was oil to fill those pipelines, because during World War II, the Allies controlled 86 percent of the world’s oil supply.
To obtain markets for finished industrial goods, and to obtain raw materials, chiefly minerals, for building armaments, and to supply their industrial complex, Germany began to look hungrily at adjacent territories. After the Franco-Prussian War of 1871, Germany annexed iron-rich Alsace-Lorraine. Unfortunately, they later found that the boundaries they set did not include the bulk of the iron deposits because of the geologic structure there. Germany captured the surface outcrop area, but the iron-formation beds, with most of the iron ore, dipped westward into French territory. They apparently had a good military department, but a poor geology department. Germany fought the First World War in part “to correct the error of 1871.” After losing World War I, however, Germany had to give up this territory.
The experience of World War I made both the Allies and the Central Powers (Germany and its allies) keenly aware of the need for minerals with which to conduct military operations. Immediate and extensive post-war mineral exploration and development programs for both foreign and domestic sources were started by Britain and France. Because these countries, especially Great Britain, still had extensive colonial holdings, there was a lot of territory to explore. Germany, in contrast, lost all its foreign possessions, which was one of the circumstances that precipitated World War II. As Germany prepared for World War II in the 1930s under Hitler, it had been unable to regain its colonies, so it began to annex its immediate neighbors who possessed useful resources. Austria was next door and had some small iron deposits. It was the first to be taken. Czechoslovakia contained famous mining districts with fairly large iron deposits, and a variety of other metals. Czechoslovakia was taken next. With the early defeat of France in World War II, Germany regained control of the iron of Alsace-Lorraine.
Growing industrialization, with its huge demand for raw materials, growing populations, and the desire for a higher standard of living were the immediate causes for the intensified search for resources. And in the background was the thought that should another war come, the materials must be available in order to survive and win it. Hitler
In World War II, once Germany had conquered France and driven the British back across the English Channel, Hitler turned east. In keeping with his belief in lebensraum (the territory necessary for national existence and economic self-sufficiency), Hitler long held the view that only the Soviet Union had adequate land and minerals to take care of Germany’s needs (Rich, 1973). After taking this region, Hitler said, “We shall become the most self-supporting state in every respect in the world. Timber we will have in abundance, iron in unlimited quantities, and the greatest manganese-ore mines in the world, and oil—we shall swim in it.” Hitler turned his armies toward the Urals and the Ukraine. This region, along with the Donets Basin, contains extensive deposits of hematite (high grade iron ore), excellent coking coal, and limestone—the three fundamental ingredients for steel-making.
Oil to move implements of war It was not only metals that became so clearly evident as important tools of warfare in World War I. Energy, chiefly in the form of that relative newcomer, oil, was obviously going to be very significant. Some of the Allied ships bringing troops and supplies across the Atlantic from the United States to Europe were coal-fired and some were oil-fired. The oil-powered ships were the better and faster vessels, greatly speeding delivery of both troops and equipment. Oil grew more important in warfare. Gasoline-powered tanks made their first appearance on the military scene in World War I. Airplanes, fueled by gasoline, primitive as they were, also came into the war in a limited fashion.
For the first time in a major conflict, trucks replaced horse-drawn vehicles on a large scale. Recognizing this in the time between World War I and World War II, world military establishments began to give serious consideration to oil supplies. This is why Hitler coveted the oilfields of the southern Soviet Union.
By the time Pearl Harbor broke out, all of Europe was under the domination of Hitler. Africa, Latin America, Australia, India, and even parts of China remained accessible to the U.S. The only competitive customer for the mineral exports of these vast areas was the United Kingdom. An agency called the Combined Raw Materials Board was created by the U.S. and the U.K. and a coordinated buying program was launched. It was possible to fight World War II without an actual shortage of these critical materials. But the key was the fact that we retained access to Latin America, to all of Africa, to most of Asia and to Australia. Had we been denied access to them, we would have been in trouble.
After it opened up to the rest of the world in the late 1800s, Japan resolved to become an empire, but it had very few natural resources (Abbott, 1916). The nearest significant coal and iron deposits were in northern China (Manchuria). In the early twentieth century, as an emerging Pacific power, Japan was increasingly in need of raw materials, particularly metals, coal, and oil to equip its military. Japan invaded Manchuria for its deposits of iron and coal, and later southeastern Asia for more raw materials. As the military seized such resources, it also gained materials for Japan’s expanding civilian economy.
If Japan had possessed adequate oil deposits within its own borders, the Japanese probably would not have gone to war with the United States.
However, as the war continued and the United States was gradually able to cut off oil shipments to Japan, the matter of fuel became increasingly desperate for the Japanese. In the waning days of the war, Japan resorted to “kamikaze” (Japanese for “divine wind”) action – suicide pilots who would crash their planes into the ever-closer ships of the U.S. Navy. This was in large part a matter of fuel efficiency, because the planes would be given just enough fuel to fly one way. They were not expected to return and the pilots were told so. More than 4,000 young Japanese men were sacrificed this way. It was a desperate way of stretching fuel supplies. If a plane did hit a U.S. Navy ship, this would be a highly effective use of a small amount of fuel. Some planes did get through the anti-aircraft barrages and did considerable damage, but many more kamikaze planes were shot down. Finally, there was not enough oil to keep either the Japanese navy or air force operational.
Chile and nitrates. For a long time there was a boundary question between Chile and Bolivia, but no one really cared about the exceedingly desolate northern Atacama Desert territory until nitrates were discovered there in great quantity. These were the causes of the Nitrate War. Chile declared war on both Peru and Bolivia on April 5, 1879. The Chileans were victorious and obtained all of the Atacama Desert area by the Treaty of Ancón in 1883. The victory was of great economic value to Chile. From 1879 to 1889, the duty on nitrate exports alone reached more than $557 million dollars, a very considerable sum in those days. The total value of nitrate exports in that period exceeded $1.4 billion. Nitrates have continued to be an important Chilean export, although the synthetic production of nitrates has reduced their value.
South China Sea. This is a region where oil was recently discovered, and the potential for future modest discoveries is good. The Spratly Islands dot the area and consist of about a hundred coral reefs, tips of rocks, and 21 slightly submerged landforms. The emergent land is less than two-square miles in total. Although these islands are located about 700 miles from China and only 100 miles from the Philippines, China claims a huge swathe of sea that overlaps and conflicts with the claims of other nations. Indeed, ownership in the strategic South China Sea is asserted in whole or in part by nine nation states, mainly China, which claims at least 80 percent, and Vietnam. In 1988, China and Vietnam clashed violently over the Spratly Islands, and in 2012, China and the Philippines are in a tense naval standoff over their competing claims there as well. All nine nations have set up little outposts on different rocks. Despite lacking clear ownership, both Vietnam and China have issued lease blocks to oil companies. In the meantime, oil-short China is building its navy and expanding its presence in the South China Sea. China needs much more oil as it plans to greatly enlarge its road network, increase motor vehicle production, and fuel its rapid industrialization, and transport system.
Many think that water may be the oil of the future relative to resource disputes. If so, nations like Egypt may face critical decisions about military action for its very survival, because nearly all of the water from its lifeblood, the Nile River, originates in other nations which are now building dams. Up from just 28 million people in 1960, Egypt now has 82 million and is expected to have 111 million in 2025, and 124 million in 2050. Clearly, Egypt has some critical times ahead. Thirsty and hungry people become desperate people.
Colombia has an on-going drug war. It is the world’s largest producer of cocaine, 90 percent of which goes to the United States. Oil accounts for a third of Colombia’s export earnings. Drug-running rebels have tried to cut the government’s oil income. Since 1986, they have attacked the country’s major pipelines more than 900 times. In 2001, they put a pipeline out of operation for 266 days, which cost the government nearly $600 million in lost revenue (Renner, 2002).
In the Democratic Republic of the Congo (formerly Zaire), major conflicts between rival groups over very rich deposits of cobalt, tin, copper, molybdenum, and diamonds have resulted in the killing and displacement of several million people. This has given rise to the evocative term “blood diamonds,” which have also helped spark and sustain cruel insurgencies in Angola, Liberia, Sierra Leone, and Ivory Coast.
Russia has been obtaining about 60% of its hard currency from the sale of oil and gas. Without this income, Russia would have a difficult time supplying its military with needed technological equipment such as state-of-the-art computers. Oil and gas earnings also buy grain, something in chronic short supply in Russia. Grain bolsters the civilian economy, but grain also feeds Russia’s large standing armies.
Will the Russia of the future prove to be a friendly neighbor to the western nations, or will it return to its political isolation and antagonism toward the West, using its huge coal, gas, and metal resources as weapons? The Cold War was a time when the Soviet Union was active in economic warfare against the NATO allies in many regions. The reality of Russia’s nearly complete self-sufficiency in minerals and energy minerals compared with other industrial nations will be important in future world affairs. With the largest and broadest energy and mineral resource base of any nation, Russia could do very well economically.
Oil: The United States, China, and Japan. In order, these are the three largest oil-consuming nations in the world, and their economies are vitally dependent on oil at the present time. It is doubtful that alternative energy sources can arrive in quantity or in time to replace diminishing quantities of oil. The result will be an intensified scramble for the remaining world oil reserves, in competition, of course, with all economies that use oil.
How this war for oil resources will be conducted is not entirely clear, but to some degree it is already in progress. All three countries are engaged in a worldwide search for oil, both through their oil companies and also through investments in various operations in oil regions—pipelines, refineries, and others. In the iron-mining region of northeastern Minnesota, for example, Chinese investment has reopened a mine and built a pelletizing plant (to upgrade the low-grade taconite iron ore—only 30 percent iron). They are shipping the product to China. This trend will continue, and the outcome is uncertain as we create a globalized economy. China is the 800-pound gorilla on the scene.
The cheap labor weapon. Economic warfare over the past two decades is also seen taking the place in the area of labor costs. Low-wage countries aided by free trade agreements have been able to transfer the manufacture of many products from industrialized countries, particularly the United States, to their own shores. This has created huge trade imbalances. China, in particular, has a large positive trade balance with the rest of the world, especially the United States.
Armed with U. S. dollars and other foreign currencies, China, and to a lesser extent India, have embarked on a worldwide buying spree to obtain a variety of raw materials. Particular investment targets are Canada for its metal resources; Chile for its metals; Venezuela for its oil; and Australia for its metals, natural gas, and coal. This investment strategy is intensifying, particularly by China as it also reaches into Africa for both metals and oil.
One result of low-wage competition for the United States has been a large decline in its manufacturing base. The increasing deficit in international balance of payments threatens the stability of the dollar. Dollars exported to pay for things previously produced domestically, come back to compete with the United States in the form of buying power of the cheap labor countries who use the dollar to bid against the United States for natural resources worldwide. Thanks to its trade surpluses, China has had a great inflow of dollars to use for worldwide resource acquisition.
War or Reason? Struggles for resources, especially oil, will continue as population pressures grow and resources become increasingly scarce (Tanzier, 1980). Will this worldwide increased demand for energy and minerals, compounded by the current exponential growth in population, be resolved by reason, or will the struggle result in war and anarchy as suggested in the very thought-provoking book by Robert D. Kaplan, The Coming Anarchy (Kaplan, 2000)? Around the world, reason and goodwill are in shorter supply than they should be. By myriad adjustments in lifestyles and economics, the world must adjust to the new realities of resource availability. It is clear that the future cannot supply a continually growing population with resources as we use them now.
Wilderness to World Power. The establishment of a government by a free people, and an open economic system together with a great variety of abundant and easily exploited natural resources, destined the United States to change from a three-million square mile wilderness to the wealthiest and most powerful nation the world had ever seen in less than 300 years. In terms of the total energy minerals and minerals spectrum, the United States was without equal among nations at the time the Declaration of Independence was signed. Also, the millions of acres of fertile soil in the nation’s heartland favored by a good growing season are unmatched in the world.
Part of its good fortune was that the United States was established at the right time. The U.S. emerged as a nation shortly after the Industrial Revolution. It started in Great Britain and promptly spread to Europe, and then to the United States. New inventions and new technologies developed rapidly. The technologies enabled people to extract and process important raw materials like iron ore in great quantities. The invention of the steam engine fostered the development of the railroad, which was able to haul raw materials cheaply and in great quantities to the factories and distribute finished products across the country.
This combination of the right time (during the spread of the Industrial Revolution), together with a poor but ambitious free people, and the right place (three-million square miles of virgin land with a tremendous variety and quantity of mineral resources), was responsible for producing the great economic and military might of the United States in the first three-hundred years of its existence.
Of the three factors, the great variety and abundance of mineral and energy resources was probably the most important. Without these, even a free people would have seen the industrial age largely bypass them, or else arrive much later. But the rich geological endowment of the United States shaped its destiny.
One might suggest that Canada also had the same potential as did the United States, but Canada has somewhat less conveniently deposited mineral resources. It does not have high-grade iron and coal adjacent to the inexpensive Great Lakes transportation system. Little oil was discovered until recently in eastern Canada (which is offshore), whereas there were numerous oil fields in Pennsylvania, Ohio, West Virginia, Kentucky, and Indiana where people first settled and industry was established in the United States. There is no region in Canada comparable to the prolific Gulf Coast region of the United States where the famous Spindletop oil gusher discovery was made in 1901. Canada’s major oil industry really dates only from post-World War II, and, although important, it does not rival the size and wide geographic distribution of oil fields all across the United States. Also, Canada’s northern geographic position with its hostile cold climate and the difficult terrain of lakes, bogs, swamps, and large areas of tundra underlain by permafrost delayed its development except for roughly a two-hundred mile wide strip of land adjacent to the United States.
It may be, however, that the best is yet to come for Canada. The world’s largest deposits of oil sands, for example, will be an asset for many decades to come, and large high-grade iron ore deposits remain to be exploited. The United States has already depleted its high-quality iron deposits as part of the price for its phenomenal economic growth.
Russia would miss one advantage the United States had in its rapid economic rise, a relatively small population compared to large mineral wealth. Today Russia would have to spread its geological wealth over a far greater number of people then the United States had when it rose to its affluent world position. In 1880, as the United States began to reap the advantages of the Industrial Revolution, the population was approximately 50 million. The Russian population today is about 142 million. Large mineral wealth spread over a small population has the potential of raising the standard of living. This has been clearly illustrated in such countries as Saudi Arabia and Kuwait. Russia missed a great opportunity. Now, although it does have mineral wealth, it has more people. And Russian oil production has peaked before the benefits of its oil riches have been enjoyed to any large degree by the average citizen. The United States combined its oil wealth with its world class motor vehicle industry to bring a degree of affluence and lifestyle for the average citizen, that would be difficult if not impossible for Russia to duplicate now.
For many years, the United States was the world’s dominant producer of most vital raw materials. The United States was, until 1982, the world’s largest copper producer. Until about 1950, it produced half the world’s oil. It has been the leader in molybdenum, zinc, and lead output, and it still has the largest recoverable coal reserves in the world. Following the 1859 discovery of oil, the United States was completely self-sufficient in petroleum for more than 100 years. Ultimately it was the possession of large oil resources and U. S. self-sufficiency that brought about the reversal of power between Great Britain and the United States. Until World War I, coal was the dominant energy source, and British coal mines were a major source. After World War I, oil became the major fuel on which the world depended. Britain at that time had no oil production. With the arrival of the oil age, economic power shifted to the United States. One might note that the dependence of the United States on foreign oil has substantially decreased the relative world economic strength of the United States,
By 1909, the United States was producing more oil than the rest of the world combined, and continued to do so through 1950. With the discovery of oil found all across the U.S., and the development of trucks and automobiles, soon a nationwide network of roads and service stations was established. Travel came into vogue, because oil was inexpensive. The average citizen could afford it. Thus the great travel boom arrived and the novel idea of motels spread across the country. The travel industry became an important part of the U. S. economy.
In the early and middle decades of the twentieth century, mineral and energy mineral resources in the U.S. were in seemingly endless supply. The United States provided its allies with vital energy and mineral resources first to win World War I, when it was said that “the Allies floated to victory on a sea of oil.” U.S. oil supplies again played a vital role in World War II. Both Japan and Germany lacked oil. In terms of metals, it has been said that both wars were fought from the great hole in the ground which is the Hull-Rust iron mine on the north side of Hibbing, Minnesota.
With cheap and abundant mineral and energy mineral resources, the United States enjoyed an unprecedented rapid rise to the world’s highest material standard of living and to economic and military pre-eminence.
When considering the future, as compared with the past, it is important to note that the United States achieved its industrial position and its high standard of living on abundant, cheap energy, and rich mineral resources. It took enormous energy to mine and smelt the ores to produce the metals vital to industrial development. It took vast amounts of energy to conquer the frontier and do the work needed to convert a raw wilderness into the world’s largest, most affluent society. During most of the time between 1940 and 1960, the United States enjoyed $3-a-barrel oil, natural gas costing about 15 cents a thousand cubic feet, and coal costing about $4 a ton, all available within the United States. Abundant and inexpensive energy sources and high-grade iron and copper deposits were exceedingly helpful to a young and rapidly growing nation. High-grade metal deposits take less energy to mine and smelt than low-grade deposits do.
The combination of high-grade ores and inexpensive energy combined to provide very inexpensive finished products that fostered economic growth. Conversely, as ore grade decreases, it takes more energy to produce the same amount of metal as previously. Combined with higher energy costs, the result is substantially higher finished product costs.
The United States’ peak of power may have been symbolized by its use of the ultimate energy weapon, the atomic bomb, to end World War II in 1945. At the time, the United States was the sole owner of this fearsome form of energy. It was the possession of a particular metal, uranium, within its borders that allowed the United States to arrive at this zenith of world power. Now this capability is shared by several countries, and the number is growing.
After the breakup of the Soviet Union in the early 1990s, there were those who argued that the United States stood alone as the unrivaled world power. Unlike 1945, however, in the 1990s the United States was no longer self-sufficient in its principal energy need, oil. In fact, it was importing more than half of its supplies, and lacked the ability to reverse the trend. The United States no longer controlled its economic destiny, which was partly in the hand of foreign oil producers. And the continuing imbalance of foreign trade, in which imported oil was the largest single (and growing) factor hurt the prestige and value of the U.S. dollar in world markets.
The United States rose to international economic dominance in record time, but in the process depleted many of its high-grade resources. The rich ores of the Mesabi Iron Range are gone. All the high-grade native copper mines of Upper Michigan are closed. The United States now searches for oil off the frozen north coast of Alaska and in the deep waters of the Gulf of Mexico. The U.S. is no longer nor will it ever be self-sufficient in oil again. Its oil reserves, once the largest in the world, are now dwarfed by those of several other countries. Although it consumes about 25 percent of the world’s oil, the U.S. now contains only about four percent of the estimated conventional proved world oil reserves.
The United States has changed from being an exporter of energy and mineral resources to now being a net importer on an increasingly large scale. In the process, the United States also went from being the world’s largest creditor nation to being the world’s largest debtor nation in less than 20 years. Oil imports now are the single largest item contributing to our annual balance of trade deficit. Other basic commodities are increasingly imported as the U. S. continues to decline in resource self-sufficiency.
The saga of the astonishing rise of the United States to affluence and power will never be duplicated in the world. There are no more virgin continents to exploit. The story of the growth of the United States has been a phenomenon beyond comparison. The question now is: where does it go from here?
ALASKAN OIL
No other oil fields the size of the approximately 12 billion-barrel Prudhoe Bay Field are expected to be discovered in Alaska. About 60 miles to the east of Prudhoe Bay, the geology suggests that perhaps a field or fields not as large as Prudhoe Bay might exist in a small portion of the coastal plain of the Arctic National Wildlife Refuge.
Alaska’s oil revenues are certain to decline in the long run, to the point where there will be little or no such income. If future generations of Alaskans are to be considered, then the abundant but transient oil revenues of today must be used differently. The political process has allowed this generation of Alaskans to capture benefits at the expense of future generations by plundering much of the wealth of its nonrenewable oil resource.
Of the initial 12 billion barrels of oil reserves in the Prudhoe Bay Field, seven billion have been produced. Production has peaked, and is now declining.
INDONESIA
This archipelago is the fourth-most populous nation in the world with 240 million people. Until 2009, it was a member of OPEC. Its oil deposits were developed by Shell Oil Company, which began as a trading company that actually dealt in part in seashells when Indonesia was a Dutch colony. This is how the Shell companies got their start, and they still retain the shell symbol. Indonesia was the prize the Japanese needed to keep their war machine going, and they occupied it for a time during World War II. After the war, Indonesia gained independence from the Netherlands, and continued to be an oil exporter. It has now become a net oil importer, with domestic oil demand now exceeding production.
Military spending by oil-rich countries, principally in the Middle East, has overshadowed all other investments. In the 12 years following the first Arab oil embargo in 1973, with the subsequent huge rise in oil prices and concurrent huge increase in revenues for oil producers, the Gulf nations spent more than $640 billion for military purposes. Iraq and Iran spent billions, which served only to finance an eight-year war of attrition ending in stalemate. An estimated million people were killed and many more were wounded. Without the oil money to finance the advanced weapons of war, it is probable that at least the casualty figures would have been less. What a tragic way to waste forever the proceeds from a non-renewable resource.
Now these countries have the latest state-of-the-art weaponry including all sorts of missiles, tanks, jet fighters, helicopters, warships, and other hardware. But there is a question whether or not these nations are safer or happier with all these devices of destruction. Arms shows in the Middle East have become regular events. It is big business. The judgment of history on the way in which some of the temporary oil riches are being spent is likely to be severe. The government-owned oil industry has been ineffective in using oil wealth for general social improvement.
MEXICAN OIL
Pemex is not run efficiently, and is grossly overstaffed with more than 108,000 employees. The oil revenues have not been used to raise the Mexican standard of living. Pemex provides the government with 40 percent of its revenues and pays 70 percent of the cost of running the national electric grid. Exactly where the rest of that tax revenue goes is uncertain.
The Cantarell oil field that supplies 62 percent of Mexico’s total production has peaked and is in steep decline.
Just to maintain Mexico’s oil production, the government will have to invest as much as $100 billion in the coming decade. Taxes prevent Pemex from retaining enough of its earnings to finance needed expenditures. Francisco Rojas, who ran Pemex from 1987 to 1994, referred to Pemex’s decaying infrastructure and lack of money for exploration saying, “ … we are looking at a time bomb….”
In Mexico in 1996, the Democratic Revolutionary Party (PRD) set up blockades at oil installations in the oil field areas of the southeast coast protesting the actions of Pemex, the Mexican government oil monopoly. The PRD said Pemex had contaminated farm land, had not cleaned up oil spills nor raised living standards as promised, but had diverted its oil money to political ends and into politicians’ pockets. Mexico is another example of how oil may prove to be a destabilizing influence on a country. The national oil company, Pemex, provides 40 percent of government income but production has dropped 30 percent from its peak in 2004. In 2009, there was a substantial cut in government employment (10,000 workers), increased personal and corporate income taxes ($13 billion), and reduced subsidies for things such as electric power. The country was living on a diminishing resource, and now “It’s crunch time in Mexico” (Smith, 2009). How this increased austerity will affect Mexican social and economic structures is not yet clear, but it will surely do so as it will in all countries dependent on income from nonrenewable resources.
NIGERIAN OIL
This is the largest African nation in terms of population, with 162 million people and a growth rate projected to increase its population to 237 million by 2025, and 437 million by 2050 (Population Reference Bureau, 2011). Nigeria remains beset with problems. It is rated as one of the most corrupt regimes in the world. Lack of civil control has reached the point that Shell Oil Company says it may not be able to continue operations there because of the ongoing sabotage of equipment. With corruption, lack of civil order, growing religion-based terrorism, a fast-growing population – already the largest of any African nation — the future for Nigeria is dim. A jihadist terrorist organization — Boko Haram — whose name translates from the Hausa language to “Western education is sacrilege,” and which began attacking Christian targets in 2010, is only the latest threat to Nigeria’s fragile stability. Oil revenues surely have not been used as wisely as they might have. Nigeria has about 37-billion barrels of oil reserves. And if civil order can be maintained, there are fair prospects for additional discoveries. But Nigerians themselves lack the ability to conduct oil operations, so foreign technology and investments are required. The country’s production is expected to peak about now.
When the day arrives that Nigeria is not oil rich, it may revert back to the bloody civil wars that marked the country 30 years ago. Today, even with oil riches, or perhaps because of them, corruption and civil unrest continue to plague Nigeria.
NORWEGIAN OIL
About 2% of Norway’s land is arable. There is not a drop of oil in Norway’s largely igneous rock terrain. Norway has always had to live in considerable part from the sea. Norway has used a lot of their oil revenues to keep unemployment low. Billions of dollars of petroleum revenues have been poured into what turned out to be money-losing projects in agriculture, iron mining, smelters, and fishing in order to keep people employed. This is a temporary fit, and does not build a sound economic base for the long term. Norway’s oil reserves are about 5.3 billion barrels. Production peaked in 2001 and is now in decline. The resulting decline in oil revenue will be a problem, but not so much as in countries with a high population growth rate. Norway’s population of 5.0 million has a very low growth rate of about 0.4 percent annually bringing its projected expected population to 5.6 million in 2025 and 6.6 million by 2050.
VENEZUELAN OIL
In spite of Venezuela’s rapid population growth and associated social needs, President Hugo Chavez purchased seven Russian MIGs, and a fleet of Russian attack helicopters in 2004. In 2005, he bought 100,000 Russian AK-47 rifles. Flush with the money from high oil prices, President Chavez spent over four billion dollars (equivalent) in 2005 and 2006 buying additional military equipment, making Venezuela the most heavily armed country in Latin America. Since 2005, Venezuela has signed contracts with Russia for 24 Sukhoi fighter jets, 50 transport and attack helicopters, and 100,000 more assault rifles. Venezuela also has plans to open Latin America’s first Kalashnikov factory to produce rifles in the city of Maracay.
In Venezuela in 1989, there may have been a preview of what could happen more widely when income from a depletable resource, such as petroleum or metals, declines. Oil income began to falter. The government had to change its free-spending ways that were based on abundant oil income from 1974 to 1979, when oil prices moved up very rapidly. It started budget cutting as oil prices declined. When subsidized bus fares were raised along with previously cheap gasoline prices, riots erupted in Caracas and 17 other cities. More than 300 persons were killed, 2,000 were injured, and several thousand arrested. The government had to rescind the increases. When the price of oil dropped in 2008 from $147 a barrel to less than $60, Venezuela’s oil-dependent economy was severely affected.
Venezuelan President Hugo Chavez began running into problems. In 2005, Venezuela’s oil production declined to 2.2 million barrels a day from its peak of approximately 3.7 million barrels a day several years earlier. To maintain support for his regime, President Chavez diverted more and more of the national oil income to financing growth in social programs so there was not enough capital left in the industry to keep oil production stable, much less increase it.
Venezuela’s social structure, in a country with a growing number of poor, is under severe strain. Recognizing the need to help the poor is commendable. As noted elsewhere, this diverts money needed to maintain oil production, so population and population growth are in conflict with preserving the resource that supports it. A smaller population would help considerably. But Venezuela’s population is 29 million and is projected to reach 35.4 million in 2025, and 42 million by 2050. Oil production and the income from it cannot possibly be increased proportionately. As in other oil-rich nations, oil revenues in Venezuela have not kept pace with the growth in population and the related growth in the costs of the social services created in earlier, more affluent oil income years.
Venezuela’s situation with its 29 million people and daily oil production of 2.2 million barrels, can be contrasted to Saudi Arabia with 28 million people and daily production of more than nine million barrels. Saudi oil is also higher quality and brings a better price than Venezuelan heavy crude. So Venezuela, with a slightly larger population, is on an oilincome diet less than one-fourth that of Saudi Arabia. Both countries are almost totally dependent on oil income for foreign exchange.
GEOTHERMAL ENERGY
The world’s largest geothermal electric generating plant is located at The Geysers, about 70 miles north of San Francisco. Electricity was first produced there in 1960. More than 2,200 megawatts was eventually being generated, the equivalent of two large dams, and enough to supply the electric power needs of about one and one-half million people. However, the field was apparently over-drilled, and production eventually fell to about 1100 megawatts. When geothermal energy is used for electric power generation, it is nonrenewable because eventually the reservoirs of steam and/or hot water will be depleted to the point where they are no longer capable of sustaining
electric power generation. The time to depletion is variously estimated to range from 40 to 100 years in most geothermal electric power fields. However, after being shut down over a period of many hundreds or perhaps thousands of years, the field will recover and could be used again, because the heat will still be there. It is only the hydro system that gathers the heat from fractured hot rocks and brings it to the well bore that becomes exhausted.
For this reason, geothermal energy used for electric power generation in a practical sense does not appear to be a renewable resource. There is, however, technology being tested which may modify this conclusion. In some geothermal fields, waste water is being injected back into the reservoir to see if the reservoir level and pressure can be maintained without reducing the temperature. At The Geysers field in northern California, a 65-kilometer pipeline was recently completed bringing waste water north from Santa Rosa for injection into the geothermal field. An earlier project brought water from a lake that resulted in a recovery of 68 megawatts of power and slowed the area’s pressure decline. If this technology continues to be successful, it can materially extend the life of geothermal electric generating fields. With proper management, it could make geothermal electric power a sustainable energy resource.
Worldwide geothermal electric power. Currently the installed capacity of geothermal-powered electric generating plants totals more than 10,000 megawatts. This is the equivalent of about seven to nine conventional coal-fired plants. Leading countries, in declining order, are the United States (2,685 MW); Philippines (1,970 MW); Indonesia (992 MW); Mexico (953 MW); Italy (810 MW); Japan (535 MW); New Zealand (472 MW); and Iceland (421 MW).
Renewable for space heating? When used for space heating, hot water usage can be controlled to be kept in balance with the natural recharge of the hydro system which brings the heat from the permeable hot rocks to the well bore. In this case, geothermal energy is a renewable resource. Thus, depending on its end use, geothermal energy can be thought of either as renewable or non-renewable. Even when used for space heating, the geothermal reservoir can become depleted if it is over-used. This seems to be the case in several of the district heating systems in southern Idaho, and at Klamath Falls, Oregon, where studies of this problem are under way. However, using geothermal water for space heating is its most efficient use and it should be pursued. The efficiency stems from the fact that there is no change from one energy form to another. When geothermal water is used to generate electric power, heat energy is changed to mechanical energy and then to electrical energy. Any change in energy form results in an energy loss (second law of thermodynamics).
The lower-temperature waters that can be used for space heating are far more abundant and widespread than the high-temperature waters required for efficient electric power generation. Even where there is no especially warm water to use, the general heat flow of the Earth can be tapped by using groundwater heat pumps, which are efficient in most temperate areas, and better in some places than standard air-to-air heat pumps.
Use of Earth’s natural heat flow It is feasible, particularly in a hilly setting, to build a split-level house with part of it below ground level. In colder countries, in particular, the natural heat flow of the Earth will add to the warmth of the house in the winter. Because it is a steady temperature, it also can have a cooling effect in the summer. Such an arrangement is a natural air conditioner. Some houses are built almost entirely underground. Whereas this requires more power for lighting, there is a net reduction in power use because both heating and cooling take far more energy than lighting does. It is also land efficient in cases where people grow gardens on the roofs of such dwellings. By using the natural heat flow of the Earth in this fashion, geothermal energy is renewable,
BIOMASS & WOOD
The exploitation of wood and other biomass is removing vegetation in many areas beyond the replacement rate, causing large and fatal landslides, devastating floods (as in Bangladesh, 1988), and widespread erosion and loss of valuable topsoil (Pimentel and Krummel, 1987
In Haiti, which was once nearly all forested, only two percent of the land is now, because the demand for charcoal, an important fuel for cooking and household use, far exceeds the reforestation rate
In the countryside around Bogota, Colombia, the highlands of Peru, and throughout much of India, Pakistan, Bangladesh, Nepal, and other parts of Asia including China, in Central America (Guatemala, Mexico, Honduras), and especially in Africa there is a serious firewood supply problem. Depleting forests and brushy groundcover has harmed the soil, both by erosion and from the loss of biomass important to soil health. It is doubtful that the use of wood for fuel can be significantly expanded. In many regions, the forests need to grow back to prevent further erosion and floods, which are already severe. Loss of trees and vegetation is a particular problem in the foothills of the Himalayas where forests once regulated the gradual run-off of water. That has been changed by deforestation and now great floods occur in the lowlands to the south, especially in the densely populated lowlands of Bangladesh. In both India and Bangladesh, deforestation has also resulted in eroded soil filling reservoirs far faster than was projected for dam sites.
Wood is still a useful local fuel for cooking and heating. Some utility companies use wood in small amounts for power generation. However, the woodlands of the world are so important to the health of the environment for preventing erosion, as a sink for carbon dioxide, and in countless other ways that they cannot be used as a sustainable fuel supply in any significant amount.
Ethanol
Dr. T. Stauffer, a research associate at Harvard, said, “The bottom line is that using alcohol to stretch gasoline is like using filet mignon to stretch hamburger.
“Renewable power and fuels will be more expensive than the dirtier sort for the ‘foreseeable future.’ This leaves the clean-energy business largely dependent on government handouts.” (The Economist, 2007).
Although ethanol is commonly thought of as a farm product, it is largely a product of the energy inputs noted, and the oil-based pesticides and fertilizers used to enhance the growing. This fact is conveniently ignored by politicians and others who endorse ethanol as an alternative to gasoline at the same time they berate the oil industry that provides the basis for modern agriculture, including corn production.
Consumer Reports (October, 2006) weighed in on the ethanol issue with a cover article The Ethanol Myth pointing out that because of the lower-energy content of ethanol (27 percent less than gasoline) ethanol may not save as much energy as claimed, and that it greatly reduces the mileage range of cars. In one test case, it reduced car mileage from 450 miles to 300 miles per tank of fuel.
Pimentel and Patzek (2005) estimate that if the entire U.S. corn crop were used to make ethanol, “ … it would replace only 6% of fossil fuel used in the U.S. And because the country has lost over a third of its agricultural topsoil, no large increase in the corn crop is possible.” Pimentel and Patzek (2005) further write: Moreover, the environmental impacts of corn ethanol are enormous. They include severe soil erosion, heavy use of nitrogen fertilizer and pesticides, and a significant contribution to global warming. In addition, each gallon of ethanol requires 1700 gallons of water (mostly to grow corn) and produces 6 to 12 gallons of noxious organic effluent.
Using food crops such as corn to produce ethanol also raises major ethical concerns. More than 3.7 billion humans in the world are currently undernourished, so the need for grains and other foods is critical. Growing crops to provide fuel squanders resources; better options to reduce our dependence on oil are available. Energy conservation and development of renewable energy sources, such as solar cells and solar-based methanol synthesis, should be given priority.
In terms of gasoline, Smil (2010) estimates that if the entire U.S. corn crop was converted to that use, it would produce an equivalent of less than 15 percent of current U.S. annual consumption.
The impossibility of using methanol as a replacement for oil in the United States is summarized by Pimentel: “If methanol from biomass (33 quads) were used as a substitute for oil in the United States, from 250 to 430 million hectares of land would be needed to supply the raw material. This land area is greater than the 162 million hectares of U.S. cropland now in production” (Pimentel, 1995).
Biodiesel also has limitations beyond either gasoline or ethanol. The Minnesota Legislature passed a law in 2005 requiring diesel sold in that state to contain 2% biodiesel. This worked fine in the summer, but given the nature of the Minnesota winters, it turned out that even 2% biodiesel in the fuel would congeal and plug the fuel line in cold weather. This aroused the ire of the truck drivers, and the law was temporarily suspended. “It’s not like they weren’t warned” said C. Fords Runge, a University of Minnesota economist who prepared a biodiesel study in 2001 on behalf of a Minnesota trucker trade group (Meyers, 2005). Others had cautioned that there would be problems. “I’d stop short of saying, ‘Ha, I told you so,” said Russell Sheaffer, Vice President of Cummins Power, a regional distributor of Cummins [diesel] engines (Meyers, 2005). B100 (100 percent biodiesel) is not recommended for use in temperatures below 40 F. Decreasing temperatures below 40 F require that increasing amounts of petroleum-derived diesel be blended into the fuel because petroleum-derived diesel does not have such temperature limitations.
LOSS OF SOIL
Overall, one-third of the topsoil of U.S. cropland has been lost over the past 200 years. Worldwide, degradation of agricultural land is causing irreversible loss of an estimated area of six million hectares (nearly 15 million acres) annually. Soil is being lost from land areas 10 to 40 times faster than the rate of soil renewal, endangering future human food security.
ENDLESS GROWTH ON A FINITE PLANET
The concept of sustainable growth is analogous to the concept of perpetual motion — both happy illusions that are detached from reality. The former ignores limitations of a finite Earth, and the latter ignores the second law of thermodynamics.
Growth in population creates a tremendous impediment to any program to solve the world’s energy problem. The nearly 80 million people being added each year to world population is more than the combined population of The Netherlands and France.
“The gain in realistic energy conservation efforts would be nullified within a decade by population growth.” Keep this statement in mind whenever a serious discussion of energy problems comes up. It is almost always overlooked.
The terms “smart growth” or “managed growth” seems to make people feel better, and they believe the problem of growth is being solved. But growth is growth by whatever name it is called. Smart or managed growth is analogous to moving the deck chairs on the Titanic closer together, and finally stacking them on top of one another. This simply results in the ship going down in a more organized manner than it would otherwise.
Herman Daly is one of few economists who understands the limits of a finite Earth. As early as 1987, in “The Steady-State Economy: Alternative to Growthmania” he related his concerns about “growthmania” and the ecosystem with the observations: The economy grows in physical scale but the ecosystem does not…. Standard economics does not ask how large the economy should be relative to the ecosystem, but this is the main question posed by steady-state economics…. Standard economics…is indifferent to the scale of aggregate resource use. In fact, it promotes an ever-expanding scale of resource use in appealing to growth as the cure for all economic and social ills…steady-state economics stresses the optimum scale of resource use relative to the ecosystem.
“Growthmania” (In growth we trust.) is the current guiding creed of both industry and government and of both main political parties in the U.S.. Chambers of commerce enthusiastically and unanimously promote it. This century will show it to be a false pursuit, even a delusion.
Abernethy (1993) makes the important observation that the carrying capacity of a region is not constant: Life support systems deteriorate from overuse and are less able to support life. This means that overpopulation in one period decreases the future number of people who can be maintained without aggravating the damage. The carrying capacity does not remain constant. It shrinks.
Nor is the solution recycling because 100% recovery cannot be achieved. Some is inevitably lost in the process. Furthermore, to recycle materials requires energy.
One of the most difficult problems is how to stabilize population size, and at what level? The issue of population size is frequently ignored, and when it is considered, it is treated very cautiously because it involves delicate cultural, religious, and racial matters. But some population size must be assumed to rationally describe what resources and technologies would be required to sustain humans indefinitely.
Pimentel and Pimentel (1979) and others at Cornell University have made perhaps the most comprehensive and quantitative study of “sustainability,” relating it to the two most vital parts of our resource base — soil and water. “For erosion under agricultural conditions, we should be losing less than 1 ton per hectare per year for sustainable farming. For groundwater resources, we should be pumping no more than 0.1 percent of the total aquifer for use.” But, we rarely manage these two vital resources based on these criteria. Pimentel et al. (1994), have suggested a figure of about two billion as the sustainable population of the Earth, and about 100 million for the United States.
The term “sustainable development” is commonly used with optimism that implies the growth trends we now experience can really be indefinitely sustained. Economists, in particular, are very fond of it. So are real estate developers and chambers of commerce. To meet objections about the continuing growth of various projects, the term “smart growth” has been invented and is widely used.
However, as Bartlett (1997a) pointed out: “The claim is made that growth management will save the environment. Whether the growth is smart or dumb, the growth destroys the environment.
Problems relating to “sustainability” come in many forms. Land use and food production are important issues. From 1980 to 2002, the U.S. per capita agricultural land declined from 1.5 acres to one acre. At the same time, population increased by 60 million. For the first time, the United States became a net importer of agricultural products (Hartmann, 2006).
In the U.S., cities occupy only 3% percent of the land area, but 26% of the best agricultural land.
We talk about energy conservation as a way to balance demand and supply. But, the goal is constantly overwhelmed by population growth. California is a good example. “Per capita energy use dropped 5% between 1979 and 1999. However, during that same 20 years the state’s population grew 43% largely as a result of immigration” (Hartmann, 2006). In spite of the per capita reduction in energy use (conservation), the amount of energy used in California increased by 93%.
As the British writer, Bligh, has stated: “Contraception is so much kinder than starvation and genocide.
Professor James Duguid (2004) from Scotland has made this observation on individual nation sustainability: The guiding ethical principle should be that each nation should live within the resources of its own country, with only such trade in surpluses as is beneficial to all. None should depend on foreign aid, on large imports, on exporting surplus people, or in otherwise appropriating the biocapacity of other countries.
In their book, The Lessons of History (1968), Will and Ariel Durant wrote: “In the last 3,421 years of recorded history only 268 have seen no war … . The causes of war are the same as the causes of competition among individuals: acquisitiveness, pugnacity, and pride, the desire for food, land, materials, fuels, mastery
Many people now living will see the year 2050. The twentieth and twenty-first centuries, in their beginning and ending, will be vastly different from any other back-to-back centuries the world has seen, or probably ever will see. Just as those alive in 1900 could not have foreseen the changes that occurred in the next 50 years, we cannot foresee the changes that will occur in the next 50 years. But it is safe to say that energy and population will be among the most prominent things to change. Perhaps the most difficult thing to visualize is what sort of social structures will arise in response to the new resource and energy paradigm. What we do know is the changes are likely to be profound. Because increasing depletion is a new paradigm, history offers no guidance on the future.
Miscellaneous
Some cities have efficient sewer systems, some do not. Forty percent of world population has no access to sewers, and millions of gallons of raw sewage are put daily into the remaining already degraded wetlands and rivers, sometimes called the Earth’s kidneys. They can no longer absorb it all. The scale of this universal human pollution problem is beginning to grow beyond what nature can effectively recycle back into the environment (George, 2008).
The rapid rise in jet fuel and gasoline prices during 2004-2007 in the U.S. was exacerbated by hurricanes damaging and destroying offshore drilling and production platforms and by reduced refinery output.
Asphalt paves 94 percent of the roads in the United States. This is a total of about four million miles, and is the basis of our entire ground transport infrastructure, except for railroads.
The pre-colonial famines of Europe raised the question: ‘What would happen when the planet’s supply of arable land ran out?’ We have a clear answer. In about 1960 expansion hit its limits and the supply of unfarmed, arable lands came to an end. There was nothing left to plow. What happened was that grain yields tripled. The accepted term for this strange turn of events is the green revolution, though it would be more properly labeled the amber revolution, because it applied exclusively to grains — wheat, rice, and corn. Plant breeders tinkered with the architecture of these three grains so that they could be hyper charged with irrigation water and chemical fertilizers, especially nitrogen…. This innovation meshed nicely with the increased ‘efficiency’ of the industrialized factory-farm system [powered with fossil fuel]…. The way in which the green revolution raised that grain [production] contributed hugely to the population boom, and it is the weight of the population that leaves humanity in its present untenable position…. All together the food-processing industry in the United States uses about ten calories of fossil-fuel energy for every calorie of food it produces.
WIND POWER devices are unsightly, noisy, kill birds, and like solar collectors, deteriorate and have to be replaced with more materials mined from the Earth. Both wind farms and solar farms need access roads to service the equipment and the motor vehicles to do it. In brief, “there is no free lunch” in the use of any alternative energy source with respect to the environment. All have an impact.

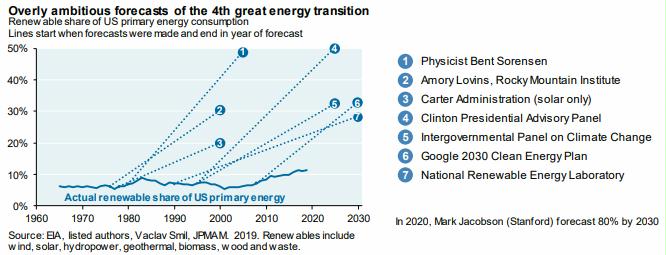



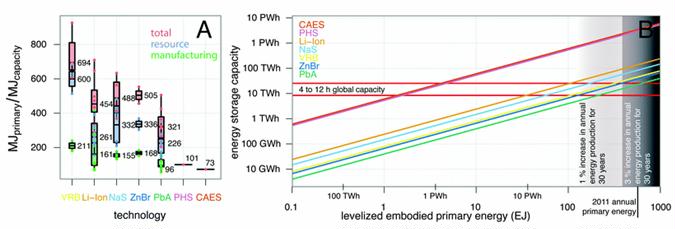
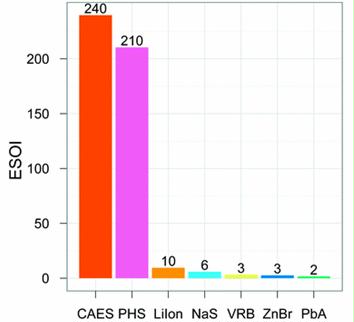
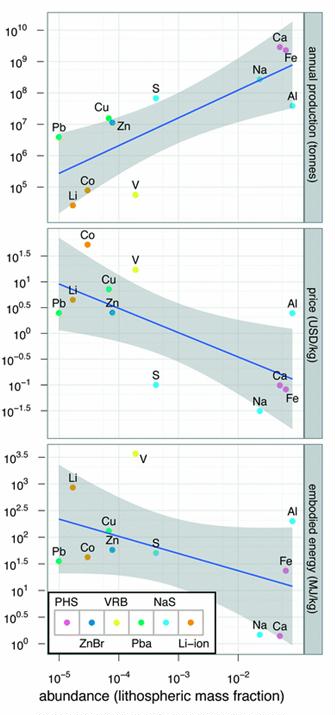
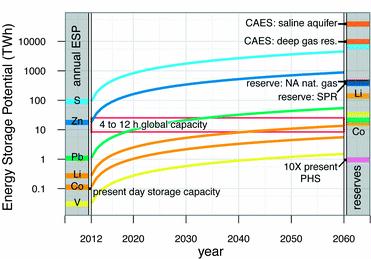
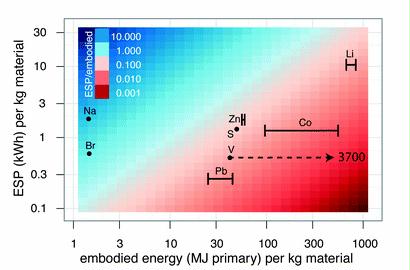
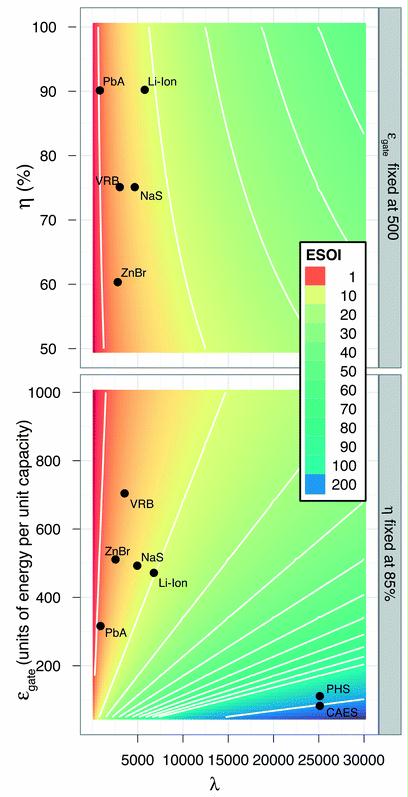
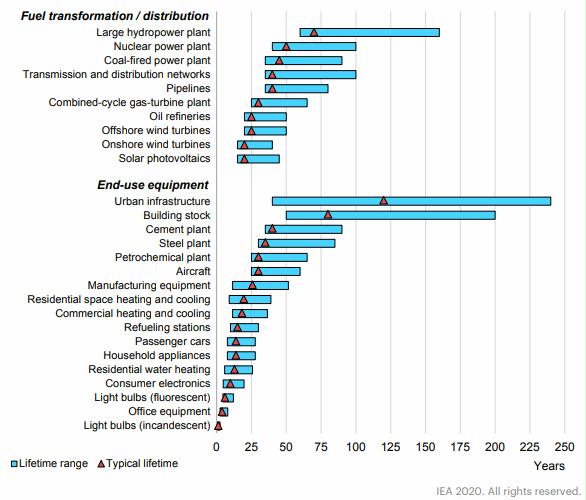
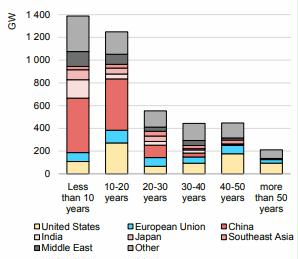
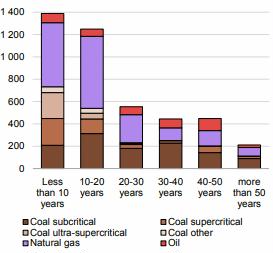
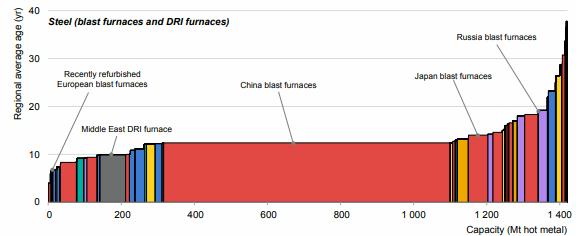
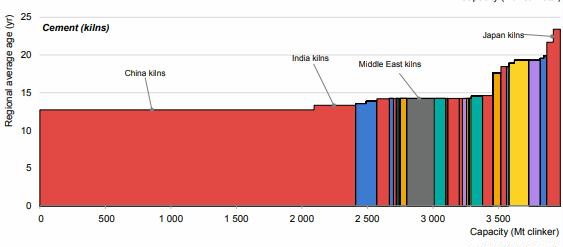

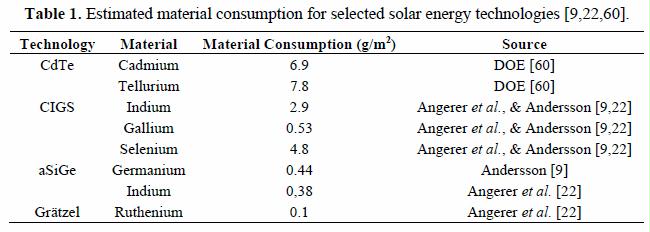
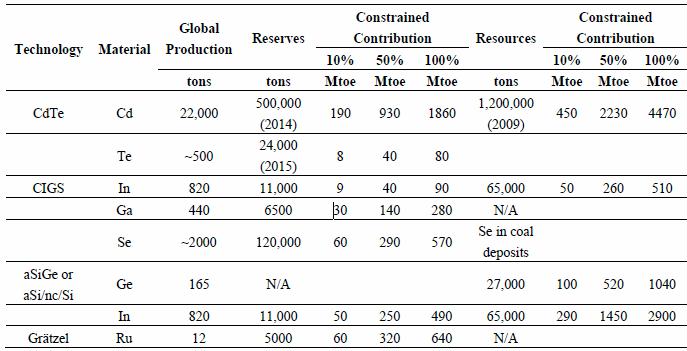
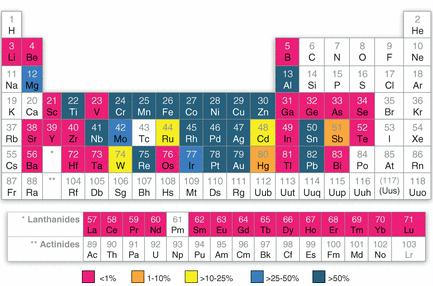
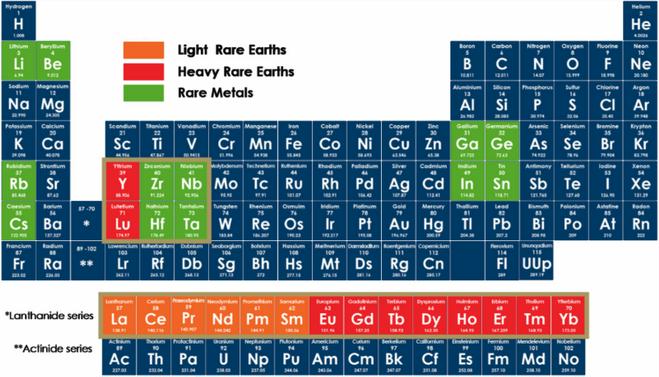
 Preface. Sunshine may be free, but the materials to make solar contraptions sure aren’t. Since sunshine arrives in a diluted form, vast expanses of solar photovoltaic panels will be needed to produce the world’s 24,000 Terawatt hours of electrical power that are now mainly generated by highly energy dense fossils such as oil and coal. Some people have taken as tab at how much land:
Preface. Sunshine may be free, but the materials to make solar contraptions sure aren’t. Since sunshine arrives in a diluted form, vast expanses of solar photovoltaic panels will be needed to produce the world’s 24,000 Terawatt hours of electrical power that are now mainly generated by highly energy dense fossils such as oil and coal. Some people have taken as tab at how much land: